Rani Therapeutics Holdings, Inc. (“Rani Therapeutics” or “Rani”) (Nasdaq: RANI), a clinical-stage biotherapeutics company focused on the oral delivery of biologics and drugs, today announced new pharmacokinetic data from a preclinical study evaluating a GLP-1, GIP and glucagon receptors incretin triagonist with a delivery method mimicking the RaniPill route of administration. A previous study with this incretin triagonist delivered transenterically demonstrated pharmacodynamic effects comparable to subcutaneous injection. Rani also previously completed a study demonstrating oral delivery of GLP-1 receptor agonist with high bioavailability via the RaniPill capsule.
“The pharmacokinetic data announced today combined with the previously announced pharmacodynamic data further highlight the RaniPill’s potential to serve as a novel delivery platform for incretin triagonists, a next generation modality for the treatment of obesity,” said Talat Imran, Chief Executive Officer of Rani Therapeutics. “Overall, the totality of RaniPill preclinical data presented to date is promising and provides scientific validation for the potential of the RaniPill to replace painful injections with oral alternatives with differentiated dosing flexibility for multiple obesity drug candidates. Looking ahead, we are focused on execution of a Phase 1 clinical trial for RT-114, a RaniPill capsule containing ProGen’s GLP-1 / GLP-2 dual-agonist, PG-102, and we are evaluating options to move forward with one or more additional molecules in the obesity space.”
Data Highlights
The preclinical study evaluated the pharmacokinetic (PK) and pharmacodynamic (PD) profiles of an incretin triagonist (GLP-1, GIP, glucagon receptors) when delivered via an endoscope-guided transenteric administration to mimic the RaniPill route of administration, versus the traditional administration route of subcutaneous (SC) injection. The study was conducted in canines separated into two groups. In Group 1 (N=3), 0.12 mg/kg of drug was administered via transenteric delivery by endoscope. In Group 2 (N=5), 0.12 mg/kg of drug was administered by SC injection. Blood samples were collected over 2 weeks for analysis of serum drug concentrations and various PD and safety biomarkers.
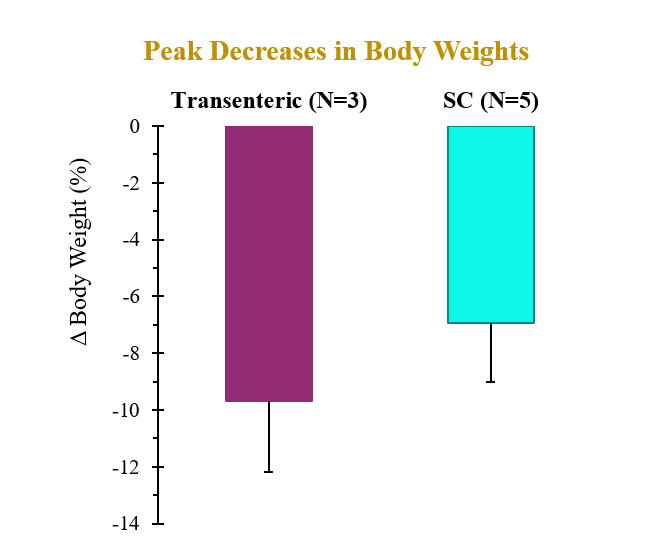
A single dose of drug delivered via either transenteric or SC routes elicited rapid decreases in body weight and serum lipids. Weight loss observed following transenteric delivery was 9.7 ± 2.5 % versus 6.9 ± 2.1 % following SC injection and is believed to be due to early satiety leading to reduced caloric intake. Additionally, the bioavailability of drug delivered via the transenteric route was comparable to that of drug delivered via the SC route at the same dose. The drug was well tolerated in both groups with no serious adverse events (SAEs) observed or changes in safety markers examined.
Transenteric delivery via the RaniPill yielded 80% relative bioavailability versus SC and differences between PK parameters (AUC, Cmax, Tmax) were not statistically significant.
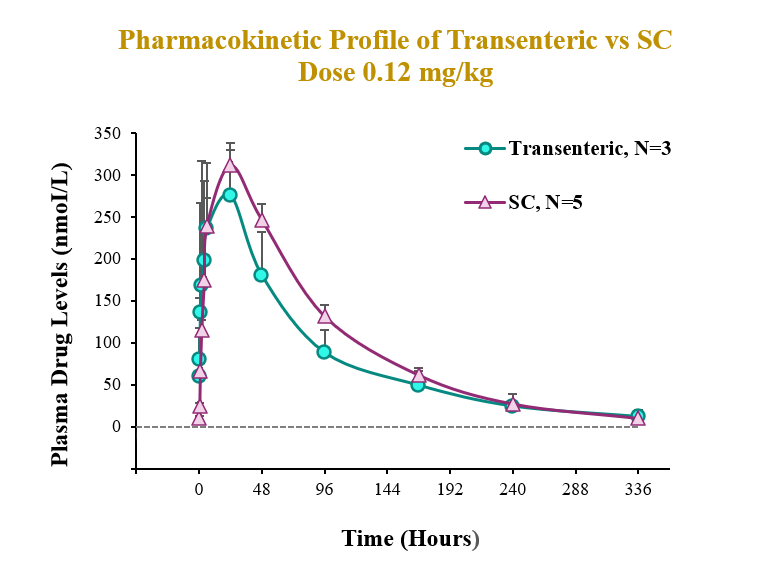
| AUC (nmol*h/L) | Cmax (nmol/L) | Tmax (h) | |
| Transenteric | 26963.3 ± 5520.0 | 334.3 ± 32.8 | 16.7 ± 10.4 |
| SC | 33509.5 ± 1576.1 | 311.5 ± 26.1 | 24 ± 0.0 |
About Rani Therapeutics
Rani Therapeutics is a clinical-stage biotherapeutics company focused on advancing technologies to enable the development of orally administered biologics and drugs. Rani has developed the RaniPill® capsule, which is a novel, proprietary and patented platform technology, intended to replace subcutaneous injection or intravenous infusion of biologics and drugs with oral dosing. Rani has successfully conducted several preclinical and clinical studies to evaluate safety, tolerability and bioavailability using RaniPill® capsule technology. For more information, visit ranitherapeutics.com.
